Get through and exclusive solution for CBSE Class 10 Science Question Paper 2019 here. Students may download the complete solution in PDF and get the correct answers for all questions given in CBSE Science question paper.
CBSE Class 10 Science paper was conducted on 13 March, 2019. Here, we are providing the detailed and appropriate answers to all the questions asked in the Class 10 Science Question Paper 2019. All the solutions have been explained as per the CBSE marking scheme that is followed while evaluating the answer sheets. Students will find it easy to understand the concepts used in the solutions.
Time allowed: 3 hours
Maximum marks: 80
General Instructions
- The question paper comprises five sections, A, B, C, D and E. You are to attempt all the sections.
- All questions are compulsory.
- Internal choice is given in Sections B, C, D and E.
- Questions number 1 and 2 in Section A are one mark questions. They are to be answered in one word or in one sentence.
- Questions number 3 to 5 in Section B are two marks questions. These are to be answered in about 30 words each.
- Questions number 6 to 15 in Section C are three-marks questions. These are to be answered in about 50 words each.
- Questions number 16 to 21 in Section D are five-marks questions. These are to be answered in about 70 words each.
- Questions number 22 to 27 in Section E are based on practical skills. Each question is a two marks question. These are to be answered in brief.
CBSE Previous Year Question Papers Class 10 Science 2019 Delhi Set I
Section - A
Question 1.
What is the function of a galvanometer in a circuit? [1]
Answer:
Galvanometer is a device that detects the presence of current in a circuit. It is also used for measuring the amount of current in the circuit.
Question 2.
Why is biogas considered excellent fuel? [1]
Answer:
Biogas is considered as an excellent fuel because:
- It causes no pollution, as it is environmental friendly.
- Biogas plant from which biogas is produced serves as an excellent way of waste disposal.
- It is economical and produces a large amount of heat per unit mass.
- MCQ Questions for Class 10 Maths with Answers PDF Download
- Most Important MCQ Questions for Class 10 Science with Solutions for 2020 Board Exam
- CBSE Previous Year Question Papers for Class 10 and 12 Last 10 Years
- CBSE Class 10 Mid Term Exam Question Papers and Sample Papers
- MCQ Questions for Class 10 Social Science with Answers PDF Download
2020 Board Exam: Download CBSE Sample Papers for Class 10 Bundle PDF |
|
|---|---|
| CBSE Sample Papers for Class 10 Maths | CBSE Sample Papers for Class 10 Standard |
| CBSE Sample Papers for Class 10 English | CBSE Sample Papers for Class 10 Sanskrit |
| CBSE Sample Papers for Class 10 Social Science | Formula Handbook for Class 10 Maths and Science |
Section - B
Question 3.
How it can be proved that the basic structure of the Modem Periodic Table is based on the electronic configuration of atoms of different elements?
OR
The electronic configuration of an element is 2, 8, 4. State its:
(a) group and period in the Modern Periodic Table.
(b) name and write its one physical property. [2]
Answer:
(a) Modern periodic law states that the physical and chemical properties of an element are the periodic function of the atomic number of that element.
Electronic configuration of the elements play an important role in the placement of elements in the modem periodic table. The valence shell electron of an element decides its position in a particular group or period for example: if the configuration of an element is 2, 1 it means that the
Li = 2, 1
It belongs to the 2nd period and 1st group.
OR
(b) Si = 2, 8, 4
1. It means that it belongs to 3rd period and 14th group.
2. The name of element is Silicon.
It is a metalloid, (i.e. element that has properties of both metals and non-metals).
Question 4.
Write two different ways in which glucose is oxidized to provide energy in the human body. Write the products formed in each case. [2]
Answer:
Glucose is oxidised in two forms to provide energy to the body:
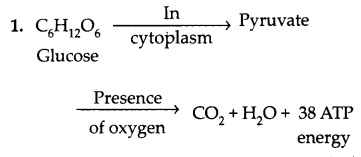
This is an example of Aerobic Respiration that happens in the presence of oxygen.
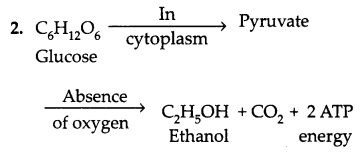
This is an example of Anaerobic Respiration.
Question 5.
Define the term power of accommodation. Write the modification in the curvature of the eye lens which enables us to see the nearby objects clearly? [2]
Answer:
The ability of the eye lens to adjust its focal length is called the power of accommodation. There should be a contraction . of ciliary muscles, that will increase the curvature of the eye lens and becomes thicker, so the focal length of the eye lens will decrease. It will thus enable us to see the objects clearly.
Section - C
Question 6.
2 g of silver chloride is taken in a china dish and the china dish is placed in sunlight for some time. What will be your observation in this case? Write the chemical reaction involved in the form of a balanced chemical equation. Identify the type of chemical reaction.
OR
Identify the type of reactions taking place in each of the following cases and write the balanced chemical equation for the reactions.
(a) Zinc reacts with silver nitrate to produce zinc nitrate and silver.
(b) Potassium iodide reacts with lead nitrate to produce potassium nitrate and lead iodide. [3]
Answer:
![]()
Initially, the colour of silver chloride is white but when it is kept in sunlight it breaks down to give silver and chlorine. Hence, the colour changes to grey.
It is an example of Photo-Chemical Decomposition.
OR
(a) The given reaction is a displacement reaction.
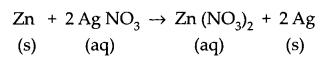
(b) It is a double displacement reaction.
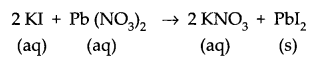
Question 7.
Identify the acid and the base from which sodium chloride is obtained. Which type of salt is it? When is it called rock salt? How is rock salt formed? [3]
Answer:
NaCl is made by reacting an acid and a base. The base is NaOH (Sodium hydroxide). So,
HCl + NaOH → NaCl + H2O
It is a Neutralization Reaction.
It is known as rock salt in crystalline form.
It is the result of the evaporation of ancient oceans millions of years ago. Sometimes pressure from deep inside the Earth forces up large masses of rocks to form salt-like domes.
Question 8.
Based on the group valency of elements write the molecular formula of the following compounds giving justification for each:
(i) Oxide of first group elements.
(ii) Halide of the elements of group thirteen, and
(iii) Compound formed when an element, A of group 2 combines with an element, B of group seventeen. [3]
Answer:
(i) Sodium is a group one element (Na) so its configuration is 2, 8, 1 and its valency is 1.
Oxide has a valency of 2.
So their formula would be
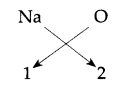
Formula for sodium oxide is Na2O.
(ii) Halide is any Halogen. Group 13 means, for example, we take Aluminium (Al) its configuration is 2, 8, 3, the valency of Al is 3, valency of Cl is 1 So,
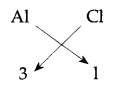
Formula would be AlCl3 (Aluminium chloride)
(iii) Element of group 2, for example, Mg 2, 8, 2 its valency would be 2.
Element of group 17 would be Cl 2, 8, 7 valency would be 1.
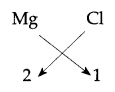
Formula would be MgCl2 - (Magnesiun Chloride)
Question 9.
Write three types of blood vessels. Give one important feature of each. [3]
Answer:
The three types of blood vessels are:
- Arteries: They carry oxygenated blood from the heart and transport it to organs.
- Veins: They carry deoxygenated blood from organs and take this blood to the heart.
- Capillaries: Arteries and veins divide to form arterioles and venules. The nutrients, hormones and gases can diffuse into the tissue cells through the walls of capillaries and vice versa.
Question 10.
Trace the sequence of events which occur when a bright light is focused on your eyes. [3]
Answer:
When bright light is focussed on our eyes it goes to the brain, the brain reverts back the message by motor neuron which contracts the pupil. The sequence of events which occur is as follows:

Question 11.
What are plant hormones? Name the plant hormones responsible for the following:
(i) Growth of stem
(ii) Promotion of cell division
(iii) Inhibition of growth
(iv) Elongation of cells [3]
Answer:
In plants, growth, development and response to the environment are controlled and coordinated by a special class of chemical substances known as phytohormones. They are synthesised in minute amounts.
(i) Gibberellins
(ii) Cytokinins
(iii) Abscisic acid
(iv) Auxins
Question 12.
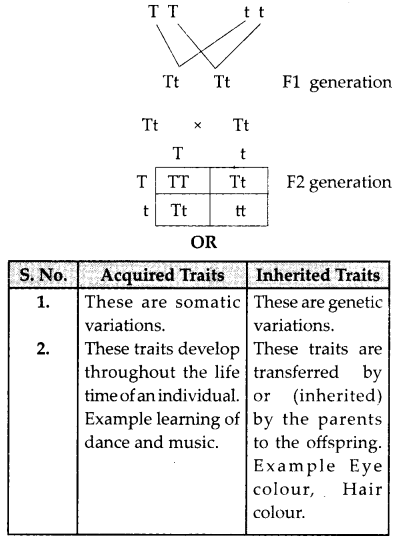
Name the plant Mendel used for his experiment. What type of progeny was obtained by Mendel in F1 and F2 generations when he crossed the tall and short plants? Write the ratio he obtained in F2 generation plants.
OR
List two differences between acquired traits and inherited traits by giving an example of each. [3]
Answer:
Mendel used pea plant (Pisum sativum) when he crossed tall and short plants the progeny obtained in FI generation were tall. When the F1 plants were selfed the F2 generations showed three tall and one dwarf plant. The genotypic ratio of F2 generation is 1 : 2 : 1 (TT : Tt : Tt : tt)
The phenotypic ratio is 3 : 1 (Tall : Dwarf)
Question 13.
What is a rainbow? Draw a labelled diagram to show the formation of a rainbow. [3]
Answer:
The Rainbow is a natural phenomenon in which the white light or sunlight splits into beautiful 7 colours by the water droplets which remain suspended in air after rain.
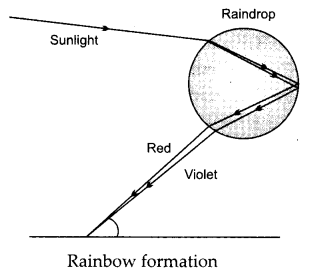
Question 14.
How can we help in reducing the problem of waste disposal? Suggest any three methods.
OR
Define an ecosystem. Draw a block diagram to show the flow of energy in an ecosystem. [3]
Answer:
The three methods by which we can reduce the problem of waste disposal are:
1. Adopting the 3R approach i.e;-Reduce, Reuse, Recycle.
2. Segregation of waste into biodegradable and non-biodegradable waste.
3. Conversion of waste into useful components like biogas and then using compost as a fertiliser.
OR
Ecosystem refers to the interaction of all the biotic and abiotic components present in a particular area. Energy flows across the trophic levels following the 10% law. Only 10% of the energy available to a trophic level is passed on to the next trophic level.
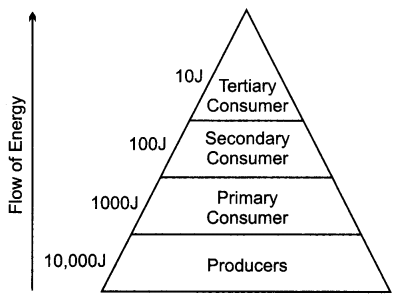
1. For example 10,000J energy is available to the producer.
2. Then 1000J energy will be available to the primary consumer.
3. 100J energy will be available to secondary consumer.
4. 10J energy will be available to tertiary consumers.
Question 15.
What is water harvesting? List two main advantages associated with water harvesting at the community level. Write two causes for the failure of sustained availability of groundwater. [3]
Answer:
Water harvesting means capturing rain where it falls or capturing the runoff in village or town. It can be done by capturing run off from rooftops and local catchments.
Advantages:
- Harvesting water allows capturing better utilization of an energy resource or when there is an immediate need.
- This water is suitable for irrigation.
Causes for failure of sustained groundwater availability are:
- Increase in agricultural activities leads to depletion of groundwater.
- Increase in population and industries are also responsible for the same.
Section - D
Question 16.
(a) List in tabular form three chemical properties on the basis of which we can differentiate between a metal and a non-metal.
(b) Give reasons for the following:
(i) Most metals conduct electricity well.
(ii) The reaction of iron (III) oxide [Fe2O3] with heated aluminium is used to join cracked machine parts. [5]
Answer:

(b) (i) Metals, for example, Na has an electronic configuration of 2, 8, 1 i.e. It has one free electron. This electron moves through the metal and conducts electric current due to the presence of free electron.
(ii) Fe2O3 (s) + 2Al (s) → Al2O3 (s) + 2Fe (l) + Heat
It is thermite reaction
This reaction is an exothermic reaction the reaction produces a large amount of heat due to which iron metal is produced in molten form and use to join the tracks.
Question 17.
Write the chemical formula and name of the compound which is the active ingredient of all alcoholic drinks. List its two uses. Write chemical equation and name of the product formed when this compound reacts with
(i) sodium metal
(ii) hot concentrated sulphuric acid
OR
What is methane? Draw its electron dot structure. Name the type of bonds formed in this compound. Why are such compounds:
(i) poor conductors of electricity? and
(ii) have low melting and boiling points?
What happens when this compound bums in oxygen? [5]
Answer:
The name of the ingredient of the alcoholic drink is ethanol.
Its formula is C2H5OH.
Two uses of ethanol (C2H5OH) are:
1. It is used as a solvent in various industries.
2. It is also used as an antiseptic solution.
(i) Reaction of ethanol with sodium metal
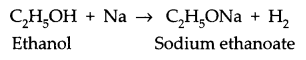
(ii) Reaction of ethanol with conc. H2SO4
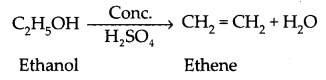
OR
Methane is a Hydrocarbon formed by the combination of carbon with Hydrogen.
Its molecular formula is CH4. It is the main greenhouse gas. The type of bond formed in methane is a covalent bond.
Electron dot structure of methane (CH4):
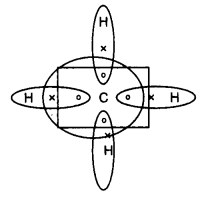
(i) Covalent compounds are poor conductors of electricity because it has no free electrons to conduct electricity. Moreover, this compound is formed by sharing of electrons. So no electron is available for the conduction.
(ii) They have weak forces of attraction between them so less energy is required to break the force of binding. Thus they have low melting and boiling points. When this compound burns in oxygen, the combustion reaction takes place.
![]()
Question 18.
Define pollination. Explain the different types of pollination. List two agents of pollination? How does suitable pollination lead to fertilization?
OR
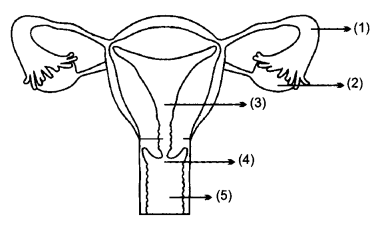
(a) Identify the given diagram. Name the parts 1 to 5.
(b) What is contraception? List three advantages of adopting contraceptive measures. [5]
Answer:
Pollination is the transfer of pollen from the anther of the stamen to the stigma of the pistil with the help of air, water and insects.
Types of pollination:
- Self-pollination: Transfer of pollen grain from the anther to the stigma of the same flower.
- Cross-pollination: Transfer of pollen grain from the anther of one flower to the stigma of other flowers of the same species.
Two agents of pollination are air, water, insects etc.
When correct species of pollen grain lands on the stigma it results in some chemical response from the ovary that causes the growth of pollen tube from pollen grain. The pollen grain slides down the pollen tube and enters the ovary where it meets the egg. This process is called as fertilization and leads to the formation of zygote.
OR
The parts of the female Reproductive system are as follows:
- Fallopian tube or Oviduct
- Ovary
- Uterus
- Cervix
- Vagina
Contraception is the method by which a female inhibits fertilization and hence presents pregnancy.
The three advantages of contraception are:
- It makes people more aware of the concept of family planning.
- It educates the people about Sexually Transmitted Diseases and ways to avoid it.
- It helps the female to space children.
- It reduces the risk of unwanted pregnancies.
Question 19.
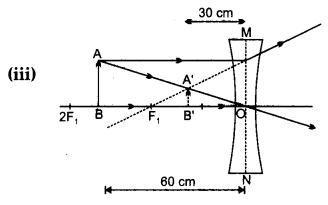
An object is placed at a distance of 60 cm from a concave lens of focal length 30 cm.
(i) Use the lens formula to find the distance of the image from the lens.
(ii) List four characteristics of the image (nature, position, size, erect/inverted) formed by the lens in this case.
(iii) Draw ray diagram to justify your answer of the part (ii). [5]
Answer:
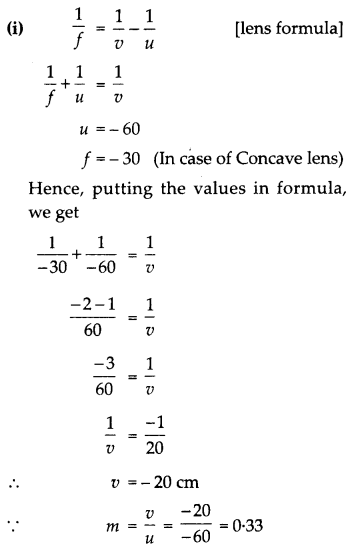
(ii) So, the image formed will be virtual, erect, small in size and image will be formed between F1 and O.
Question 20.
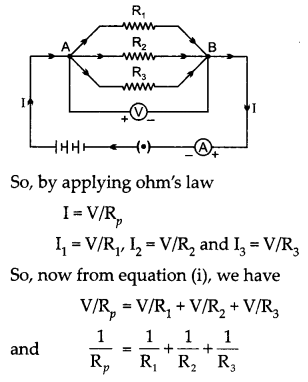
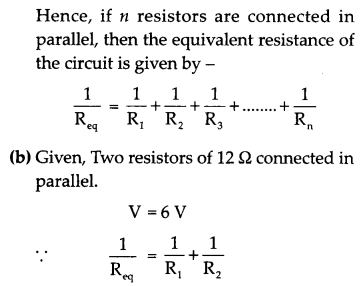
(a) With the help of a suitable circuit diagram prove that the reciprocal of the equivalent resistance of a group of resistances joined in parallel is equal to the sum of the reciprocals of the individual resistances.
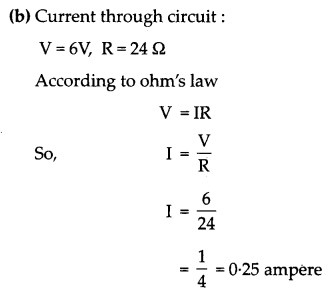
(b) In an electric circuit two resistors of 12 Ω each are joined in parallel to a 6 V battery. Find the current drawn from the battery.
OR
An electric lamp of resistance 20 Ω and a conductor of resistance 4 Ω are connected to a 6 V battery as shown in the circuit. Calculate:
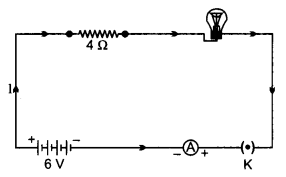
(a) the total resistance of the circuit,
(b) the current through the circuit,
(c) the potential difference across the (i) electric lamp and (ii) conductor, and
(d) power of the lamp. [5]
Answer:
(a) It is observed that total current I is equal to the sum of separate currents.
I = I1 + I2 + I23 …(i)
Let Rp be the equivalent resistance of the parallel combination of resistors.


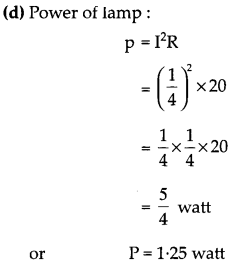
Question 21.
What is a solenoid? Draw the pattern of magnetic field lines of (i) a current-carrying solenoid and (ii) a bar magnet. List two distinguishing features between the two fields. [5]
Answer:
The solenoid is a long cylindrical coil of wire consisting of a large number of turns bound together very tightly.
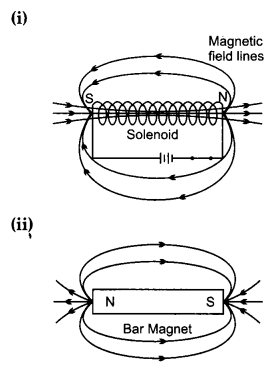
Distinguishing features are as follows:
- Magnetic field outside the solenoid is negligible as compared to the bar magnet.
- Magnetic field of solenoid can be varied as per our requirement just by changing current or core of solenoid but in bar magnet it is fixed.
Section - E
Question 22.
Blue litmus solution is added to two test tubes A and B containing dilute HC1 and NaOH solution respectively. In which test tube a colour change will be observed? State the colour change and give its reason.
OR
What is observed when 2 mL of dilute hydrochloric acid is added to 1 g of sodium carbonate taken in a clean and dry test tube? Write the chemical equation for the reaction involved. [2]
Answer:
Test tube A will show colour change because acids turn blue litmus to red. So the final colour of the test tube would be red.
OR
A brisk effervescence would be observed if HCl is added to Na2CO3 due to the evolution of CO2 gas.
Na2CO3 + HCl → NaCl + H2O + CO2.
Question 23.
In three test tubes A, B and C, three different liquids namely, distilled water, underground water and distilled water in which a pinch of calcium sulphate is dissolved, respectively are taken. An equal amount of soap solution is added to each test tube and the contents are shaken. In which test tube will the length of the foam (lather) be longest? Justify your answer. [2]
Answer:
Length of foam would be longest in test tube A. It is because it does not have any ions, so the length of the foam would be longest.
Question 24.
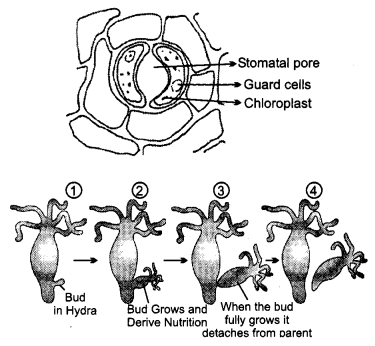
A student is observing the temporary mount of a leaf peel under a microscope. Draw a labelled diagram of the structure of stomata as seen under the microscope.
OR
Draw a labelled diagram in the proper sequence to show budding in hydra. [2]
Answer:
Question 25.
In the experimental set up to show that “CO2 is given out during respiration”, name the substance taken in the small test tube kept in the conical flask. State its function and the consequence of its use. [2]
Answer:
The chemical kept in the test tube is KOH-Potassium Hydroxide. The function of this chemical is to absorb the vapours of CO2.
Question 26.
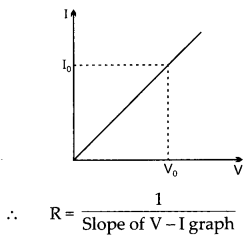
While studying the dependence of potential difference (V) across a resistor on the current (I) passing through it, in order to determine the resistance of the resistor, a student took 5 readings for different values of current and plotted a graph between V and I. He got a straight line graph passing through the origin. What does the straight-line signify? Write the method of determining the resistance of the resistor using this graph.
OR
What would you suggest to a student if while performing an experiment he finds that the pointer/needle of the ammeter and voltmeter do not coincide with the zero marks on the scales when the circuit is open? No extra ammeter/voltmeter is available in the laboratory. [2]
Answer:
The straight line in the graph signify that potential difference and current are directly proportional to each other.
The method of determining resistance of resistor using the graph is by Ohm’s law,
V = IR and by calculating the slope from the points mentioned on the graph
OR
This is called the zero error of the scale of ammeter or voltmeter. If there is a zero error then this error is subtracted from the value that depicts when the circuit is closed otherwise accurate readings will not be recorded.
Question 27.
List four precautions which a student should observe while determining the focal length of a given convex lens by obtaining image of a distant object on a screen. [2]
Answer:
Preacutions are as follows:
- Fix the lens vertically in lens holder.
- Base of lens and white screen shoule be in a line with measuring scale.
- There should not be any obstacle in path of lens.
- Record the position of lens and screen when sharp image is formed.
CBSE Previous Year Question Papers Class 10 Science 2019 Delhi Set II
Note: Except for the following questions, all the remaining questions have been asked in the previous set.
Section - A
Question 1.
Name and define the SI unit of current. [1]
Answer:
Ampere is the SI unit of current.
1 Ampere current can be defined as a unit charge flowing per second in the circuit.
1 amp = [latex]\frac { 1 coulomb }{ 1 second }[/latex]
Question 2.
Write the name of the main constituent of biogas. Also, state its percentage. [1]
Answer:
Methane is the main constituent of biogas. Its formula is CH4.
Its percentage is approximately 50-75%.
Section - B
Question 3.
Write the name, symbol and electronic configuration of an element X whose atomic number is 11.
OR
Can the following groups of elements be classified as Dobereiner’s triad:
(a) Na, Si, Cl
(b) Be, Mg, Ca
Atomic mass of Be-9; Na-23, Mg-24, Si-28, Cl-35, Ca-40, Justify your answer in each case. [2]
Answer:
The element whose atomic number is 11 is Sodium
Its symbol is Na
Electronic configuration -2, 8, 1
Valency 1
OR
(a) Na, Si, Cl cannot be classified as Dobereiner’s triad because here the elements do not belong to the same group and have different electronic configuration.
Na - 2, 8, 1; Si - 2, 8, 4 and Cl - 2, 8, 7
(b) Be, Mg, Ca are the elements of Dobereiner’s triad because the mass of Mg is the arithmetic mean of the other two elements
i.e; Be and Ca = [latex]\frac { 9+40 }{ 2 }[/latex] = 24.
Question 4.
How is O2 and CO2 transported in human beings? [2]
Answer:
Oxygenated blood is transported in human beings by the arteries. The deoxygenated blood is carried by the veins.
Question 5.
Write the structure of eye lens and state the role of ciliary muscles in the human eye. [2]
Answer:
The eye lens of the human eye is a convex lens that is thick in the middle and thin from the edges. It converges the incident light rays and forms the image on retina.
Role of ciliary muscles:
- It changes the shape of the lens in eye to, helps with focusing.
- It helps to regulate the flow of aqueous humour in the eye.
Section - C
Question 6.
Identify the acid and base which form sodium hydrogen carbonate. Write a chemical equation in support of your answer. State whether this compound is acidic, basic or neutral. Also, write its pH value. [3]
Answer:
The acid and base that forms Sodium Hydrogen Carbonate are as follows:
Acid - H2CO3
Base - NaOH
The chemical equation for its formation will be

The compound will be basic and its pH value will be around 8.
Question 9.
Define the term transpiration. Design an experiment of demonstrating this process. [3]
Answer:
Loss of excess water from the leaves of plants with the help of stomata is called transpiration.
Transpiration Experiment
Material Required: A potted plant, A polythene, Tape
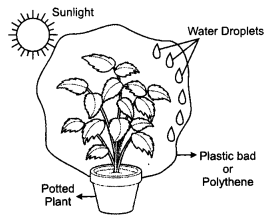
- Take a potted plant and enclose the leaves of the plant with a big plastic or polythene bag.
- Now keep the plant in sunlight for two hours.
- After some time you will be observing the water droplets on the polythene sheet.
- It depicts the loss of water from the leaves of the plant depicting transpiration.
Question 10.
What is the feedback mechanism of Harmonic regulation? Take the example of insulin to explain this phenomenon. [3]
Answer:
Hormones control many cell activities, so they are very important for homeostasis most hormones are regulated by feedback mechanisms. A feedback mechanism is a loop in which a product feeds back to control its own production. Most hormone feedback mechanism involves negative feedback loops. Negative feedback keeps the concentration of a hormone within a narrow range.
For example,
The control of blood sugar by insulin is an example of a negative feedback mechanism. When blood sugar rises, receptors in a body sense a change. In turn the control center secretes insulin into the blood effectively lowering the blood sugar level.
Question 13.
Why should there be an equitable distribution of resources? List three forces that would be working against an equitable distribution of our resources. [3]
Answer:
There should be equitable distribution of the resources so that all the people irrespective of being rich and poor have access and makes use of the resources. Equitable resource distribution also leads to sustainable development.
Three factors that work against the equitable distribution of resources are:
- Overexploitation of resources by industrialists for short-term gains.
- Improper management which has led to the access of natural raw material to a certain group of people which are either influential.
- Wastage by the people who have an indifferent attitude towards the environment, use the resources with short term aim hence leading to depletion of resources.
CBSE Previous Year Question Papers Class 10 Science 2019 Delhi Set III
Note: Except for the following questions, all the remaining questions have been asked in previous sets.
Section - A
Question 1.
If you could use any source of energy for heating your food which one would you prefer? State one reason for your choice. [1]
Answer:
I would prefer a solar cooker for heating food because solar cooker is environmental friendly and causes no pollution.
Question 2.
Write the function of the voltmeter in an electric circuit. [1]
Answer:
Voltmeter measures the potential difference across two points in a circuit. It is always connected in parallel in the circuit.
Section - B
Question 3.
What happens to the image distance in the normal human eye when we decrease the distance of an object, say 10 m to 1 m? Justify your answer. [2]
Answer:
The image distance will remain unaffected even if we change the object distance because the image is formed on the retina.
Question 4.
List two different functions performed by the pancreas in our body. [2]
Answer:
- Pancreas makes two hormones i.e., insulin and glycogen that regulates blood sugar level.
- It secretes pancreatic juice that aids in the digestion of food.
Section - C
Question 7.
List three advantages each of:
(i) exploiting resources with short term aims, and
(ii) using a long term perspective in managing our natural resources. [3]
Answer:
(i) 1. Short term exploitation will meet the demand of the present generation.
2. There will be no shortage of resources.
3. Larger aspect regarding the details of a reserve can be studied also.
(ii) 1. Long term management of natural resources would meet the demand of present and future generation.
2. No harmful impact on the environment.
3. Equitable distribution of resources amongst the rich and poor.
Question 9.
Nervous and hormonal systems together perform the function of control and coordination in human beings, Justify this statement with the help of an example. [3]
Answer:
The nervous system controls and coordinates all the functions in the body. It carries out its function in close coordination with hormonal system. Nerves don’t reach every hook and comer of body, hence needs assistance from hormones to control all the parts of body. Nervous control is faster.
Hormonal control is slower. Hormonal control is based on feedback mechansim and tells body to pace up or slow down. Nervous control is on other hand a direct control. So we can say that both system work in harmony.
Question 11.
What is photosynthesis? Explain its mechanism. [3]
Answer:
The green plants are called autotrophes. They make their own food by the process of photosynthesis. In this process the plants capture the sun rays by the pigment present in the leave called as chlorophyll. Then in the presence of carbondioxide and water they make food for the plants in the form of glucose. In this process oxygen gas is also evolved.
Equation for the mechanism:

Question 15.
Explain the following: [3]
- Sodium chloride is an ionic compound which does not conduct electricity in solid state where as it does conduct electricity in molten state as well as in aqueous solution.
- Reactivity of aluminium decrease if it is dipped in nitric acid.
- Metals like calcium and magnesium are never found in their free state in nature.
Answer:
- Sodium chloride is an ionic compound but it conducts electricity only in molten and aqueous state because in molten and aqueous form the compound liberate to give ions. These ions move freely and hence conduct electricity.
- Reactivity of aluminium decreases if it is dipped in nitric acid because it is a strong oxidising reagent. The layer of aluminium oxide prevents further reaction of aluminium due to which its reactivity decreases.
- Metals like calcium and magnesium are never found in their free states in nature because these metals are present on the top of the reactivity series. They are so reactive that they react with gases and surrounding elements, form compounds and thus are not found in free state.
Section - D
Question 17.
(a) Draw magnetic field lines produced around a current-carrying straight conductor passing through cardboard. Name, state and apply the rule to mark the direction of these field lines.
(b) How will the strength of the magnetic field change when the point where the magnetic field is to be determined is moved away from the straight wire carrying constant current? Justify your answer. [5]
Answer:
(a) The right-Hand Thumb rule is used to mark the direction of magnetic field lines produced around a straight current carrying conductor.
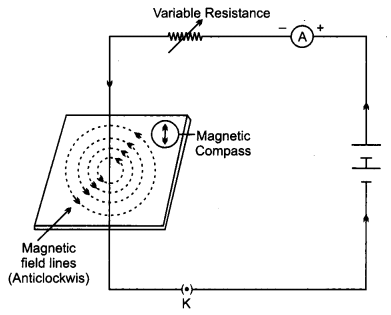
The Right-Hand Thumb rule indicates the direction of the magnetic field for a known direction of current. When the thumb is in downwards direction then the direction in ‘ which fingers are wrapped is clockwise. So magnetic field is clockwise.
(b) To determine the magnetic field, compass needle is used. On moving away from the compass needle from the straight wire, the deflection of the compass needle decreases showing the decrease in the strength of magnetic field. It happens because the strength of magnetic field produced by a straight wire at any point, is inversely proportional to the distance of wire from the point.